An Injection Every Six Months to Prevent HIV: Lenacapavir Arrives in Africa
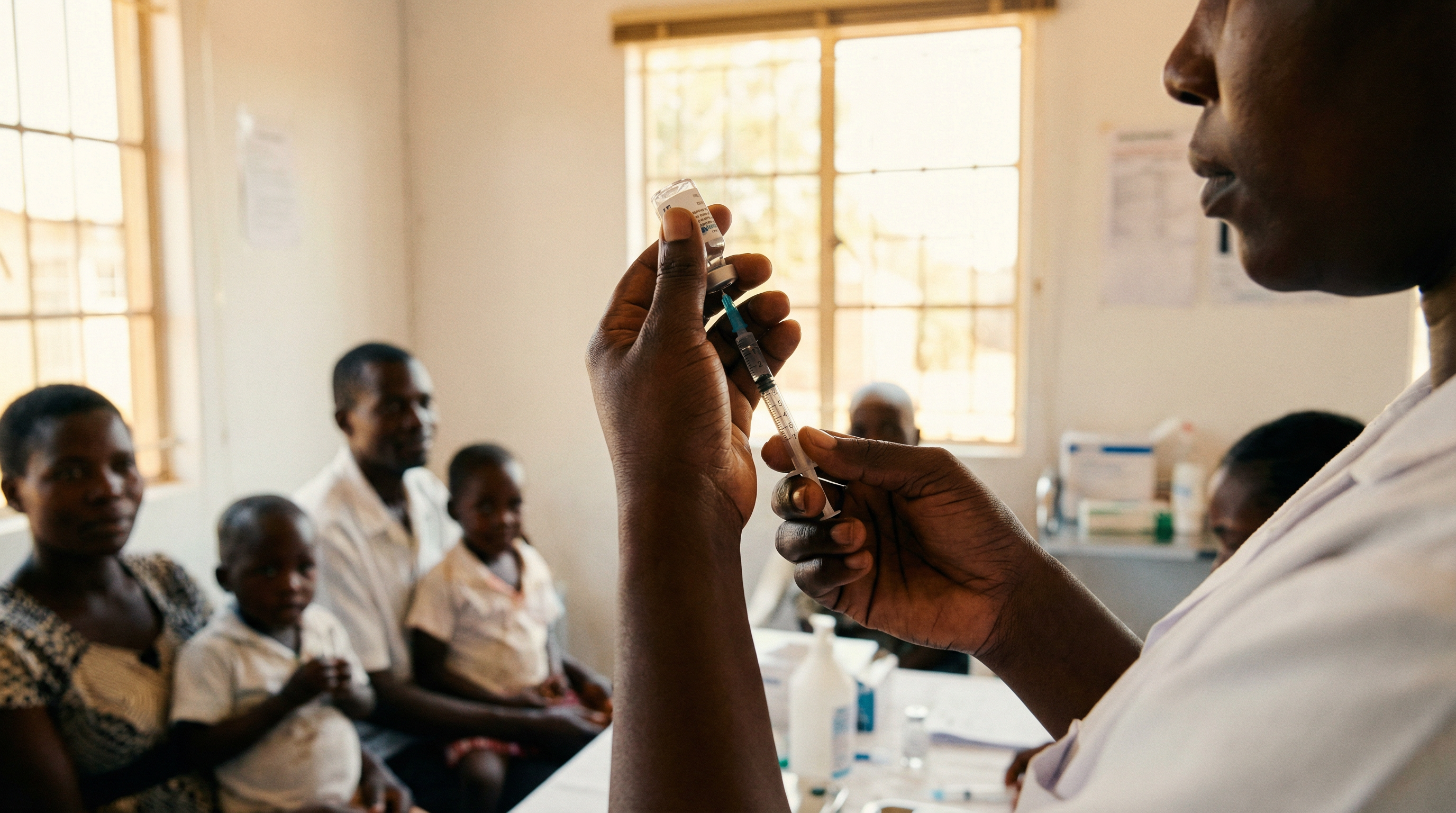
In February 2026, a clinic in Harare, Zimbabwe, administered the first lenacapavir injections to young women who came for a routine prevention appointment. Two shots per year, in the arm or abdomen, and protection against HIV is ensured for six months. No daily pill to swallow, no medicine box to hide, no routine to maintain for years. The procedure takes a few seconds. The protection lasts 180 days.
Lenacapavir, developed by Gilead Sciences, is the first long-acting injectable medication approved for HIV prevention. The PURPOSE I and PURPOSE II clinical trials, presented at the CROI conference in February 2026, confirm an efficacy of 96% in women and 88% in men. Three African countries have begun distribution. The price varies by a factor of 500 between the United States and Africa.
Forty Years of Struggle, One Million New Infections Per Year
HIV remains an active epidemic. In 2024, 1.3 million people were newly infected worldwide, according to UNAIDS. Sub-Saharan Africa accounts for 65% of these infections. Young women aged 15 to 24 represent a quarter of new infections on the continent, while they constitute only 10% of the population. Every week, 4,000 adolescent girls and young women contract the virus in Africa.
Progress is real: AIDS-related deaths have decreased by 69% since the peak in 2004, thanks to the widespread use of antiretrovirals. But the curve of new infections has stagnated since 2018. UNAIDS' goal of fewer than 200,000 new infections per year by 2030 requires an 85% reduction in six years. At the current pace, this goal will not be met. Lenacapavir arrives in this context: a potentially decisive tool to relaunch prevention that is plateauing. [5]
Adherence, Not Efficacy, is the Real Problem in Prevention
Oral PrEP (Truvada, Descovy) works. Taken correctly, it reduces the risk of infection by more than 90%. The problem is not the medication, but the daily lives of the people who must take it.
Taking a pill every day, without interruption, for years, requires a discipline that most people cannot maintain. UNAIDS data shows that adherence to oral PrEP drops after the first six months of treatment. Among young women in sub-Saharan Africa, dropout rates reach 40% to 60% in the first year. The reasons are concrete: stigma (being seen with HIV medication in a village where everyone knows each other), gastrointestinal side effects, distance from distribution centers, and simply forgetting. [5]
For an 18-year-old adolescent in Bulawayo or Kisumu, oral PrEP requires continuous effort in an environment that facilitates nothing. Lenacapavir transforms this daily effort into two annual appointments. The injection is administered by a healthcare professional, eliminating the risk of forgetting and the mental burden of adherence.
The PURPOSE Trials: Unambiguous Results
The PURPOSE I trial recruited over 5,300 adolescent girls and young women in sub-Saharan Africa. Three arms: lenacapavir by semiannual injection, F/TAF by daily pill, F/TDF by daily pill. The 52-week results, presented by Nkosiphile Ndlovu of the Wits Reproductive Health and HIV Institute (South Africa), are clear. [1]
Of the 2,000 participants who received lenacapavir, two were infected. One had missed her injection and switched to oral PrEP. The second had received all her injections on time. This case remains unexplained. In the oral PrEP arms, 77 seroconversions were recorded. The gap is massive: lenacapavir reduces the risk of infection by 25 to 30 times compared to pills, in the same populations.
The PURPOSE II trial, conducted among 2,179 men who have sex with men, confirms the trend: three infections with lenacapavir versus twelve with oral PrEP, a reduction of 88%. Two of the three infections had already been reported in the interim results. The third occurred in a participant who had received all her injections and had blood levels consistent with targets. Stephanie Cox of Gilead acknowledged during the Q&A session: "We don't know why they occurred. Only four cases out of thousands of participants." [1]
Zimbabwe, Kenya, and Nigeria Lead the Way
Deployment has been rapid. Zimbabwe began distribution on February 19, 2026, just three months after FDA approval in the United States. Kenya received 21,000 doses the same week, at a negotiated price of approximately $54 per person per year. Nigeria launched distribution at about $35 per person per year. [2] [3]
These prices are the result of negotiations led by the Global Fund and other international organizations. In October 2024, Gilead granted six voluntary licenses to generic manufacturers in Egypt, India, and Pakistan, covering 120 low- and middle-income countries. The estimated production cost of the generic is around $25 per person per year. [4]
On March 5, 2026, South Africa took a further step by launching a tender to identify a local manufacturer capable of producing lenacapavir on the continent. The initiative, coordinated by the South African National AIDS Council and supported by Unitaid, aims to bring production closer to where the need is greatest. Sub-Saharan Africa accounts for 65% of new HIV infections globally. If a South African manufacturer obtains the license, it would be the first long-acting injectable antiretroviral manufactured in Africa. [4]
$28,000 in the United States, $54 in Kenya
The price gap is the central point of tension in this story. In the United States, Gilead markets lenacapavir at a list price of $28,000 per year. In Africa, negotiated prices range between $35 and $54. The ratio is 1 to 500.
This differential reflects the pharmaceutical industry's tiered pricing model: high prices in rich countries to fund R&D, low-priced licenses for poor countries. The estimated production cost of $25 per person per year suggests that margins remain comfortable even at the African price.
But the unit price is only part of the problem. Cold chain logistics, training healthcare professionals in subcutaneous injection, and establishing semiannual monitoring systems represent concrete challenges in already strained health systems. In Nigeria, provider training was completed in February 2026, but coverage remains limited to urban centers.
PEPFAR Threatened: Prevention Funding Falters
The deployment of lenacapavir in Africa depends largely on international funding. PEPFAR, launched by George W. Bush in 2003, is the largest global health program ever created by a single country. In twenty years, it has invested over $110 billion in the fight against HIV and funds the treatment of 20.1 million people.
In 2025, the Trump administration announced a 30% reduction in funds allocated to the program. The consequences are already visible on the ground. In Mozambique, PrEP clinics have reduced their hours. In Uganda, community testing programs have been suspended. The Global Fund has launched a replenishment appeal for 2026-2028 with a target of $18 billion. If the United States reduces its historical contribution (one-third of the budget), other donors will have to compensate.
It is in this context of financial fragility that lenacapavir enters the African market. The irony is striking: the most effective drug ever developed for HIV prevention arrives at a time when the means to distribute it are threatened.
Lessons from Cabotegravir: An Instructive Precedent
Lenacapavir is not the first long-acting injectable antiretroviral. Cabotegravir (Apretude, ViiV Healthcare), approved in 2021, is administered every two months via intramuscular injection. The HPTN 083 and HPTN 084 trials showed efficacy superior to oral PrEP. But deployment has been slow.
Three years after approval, cabotegravir remains poorly distributed in Africa. The reasons are multiple: the price (over $20,000 per year in the United States, negotiations still ongoing for low-income countries), the frequency of injections (six per year versus two for lenacapavir), and the need for an intramuscular injection (more complex than the subcutaneous injection of lenacapavir, which can be administered by a community health worker after short training).
Lenacapavir learns from these obstacles. The subcutaneous injection is simpler. The semiannual frequency reduces the number of visits. And the voluntary licenses granted as early as 2024, even before regulatory approval, have accelerated availability in the most affected countries.
What Lenacapavir Does Not Solve
Lenacapavir is not a miracle solution. Four breakthrough infections despite timely injections raise an unresolved scientific question. Resistance mutations detected in infected individuals under treatment raise long-term concerns.
The drug only protects against HIV. It does not replace condoms for the prevention of syphilis, gonorrhea, or chlamydia, the incidence of which is increasing in many countries. And the US price of $28,000 per year effectively excludes a portion of the population in rich countries who do not have health insurance covering PrEP. In the United States, approximately 1.2 million people are eligible for PrEP but do not take it.
The Question of Resistance: A Risk to Monitor
The four breakthrough infections observed in the PURPOSE trials raise an important scientific question. In individuals infected despite lenacapavir, drug resistance mutations have been detected. These mutations, if they spread, could reduce the long-term effectiveness of lenacapavir.
The risk is theoretical at this stage: four cases out of thousands of participants. But the history of the fight against HIV teaches caution. Resistance to first-line antiretrovirals emerged in the 2000s, forcing a switch to more expensive second- and third-line treatments. The WHO recommends systematic resistance monitoring in countries deploying lenacapavir.
Lenacapavir acts through a unique mechanism of action: it targets the HIV capsid, a protein essential for viral replication. This target is different from that of other antiretrovirals, meaning that resistance to lenacapavir does not compromise the effectiveness of other treatments. But the reverse is also true: if lenacapavir fails, options for injectable PrEP replacement remain limited.
From Clinical Trial to Public Health
With 1.3 million new infections per year and a goal of less than 200,000 by 2030, lenacapavir will need to reach millions of people to change the trajectory of the epidemic. Clinical trials have demonstrated efficacy. The simultaneous deployment in Zimbabwe, Kenya, and Nigeria, three months after approval, is a positive signal. The South African tender for local production marks a break from the historical model where drugs are developed in the North and distributed in the South with years of delay.
The question is no longer scientific. It is logistical, financial, and political. Lenacapavir works. It remains to be seen whether health systems and international funding will be up to the task of this molecule.
Sources
- https://www.sfaf.org/collections/beta/new-research-high-prevention-efficacy-and-breakthrough-infections-on-lenacapavir-for-prep/
- _blank
- noopener


